Purpose
To test the effect of three different concentrations of HCl (0.1M, 1.0M, 2.0M) on the rate at which a piece of zinc metal dissolves.
Hypothesis
If the rate at which a piece of zinc metal dissolves is related to the different concentrations of HCl, then the higher the concentration of HCl, the faster the rate at which the piece of zinc metal dissolves.
Independent Variable: Concentration of HCl
Dependant Variable: Rate at which a piece of zinc metal dissolves
Controlled Variables: Amount of zinc metal
Beaker used
Amount of HCl
Materials
Procedure:
1. Measure out 5 mL of 0.01M Hydrochloric Acid with the Graduated Cylinder and pour it into a test tube using a funnel
2. Drop 1 strip of zinc in the test tube and record the time it takes for the zinc metal to completely dissolve
3. Dispose the Hydrochloric Acid appropriately and rinse the test tube thoroughly
4. repeat steps 1-3 for the second trial
5. calculate the mean between the two trials.
6. Measure out 5 mL of 1.0M Hydrochloric Acid with the Graduated Cylinder and pour it into a test tube using a funnel.
7. Drop 1 strip of zinc in the test tube and record the time it takes for the zinc metal to completely dissolve.
8. Dispose the Hydrochloric Acid appropriately and rinse the test tube thoroughly.
9. Repeat steps 6-8 for the second trial
10. Calculate the mean between the two trials.
11. Measure out 5 mL of 2.0M Hydrochloric Acid with the Graduated Cylinder and pour it into a test tube using a funnel.
12. Drop 1 strip of zinc in the test tube and record the time it takes for the zinc metal to completely dissolve.
13. Dispose the Hydrochloric Acid appropriately and rinse the test tube thoroughly.
14.Repeat steps 11-13 for the second trial
15. Calculate the mean between the two trials.
Discussion
In my hypothesis I stated that If the rate at which a piece of zinc metal dissolves is related to the different concentrations of HCl, then the higher the concentration of HCl, the faster the rate at which the piece of zinc metal dissolves. My predictions were proved to be correct compared to the outcome of our experiments. These predictions could be hypothesized correctly due to the research done in the BC Science 10 textbook where it clearly states that if there is a greater concentration of reactant atoms and molecules present, there is a greater chance that collisions among them will occur.
In this experiment we tested the effect of concentration of HCl or Hydrochloric acid at which a piece of Zinc Metals dissolved. All control variables, a variable that remains unchanged or held constant to prevent its effects on the outcome of the experiment, were put forth and used as to find a result with no possible sources of error with a pure and unaltered result.
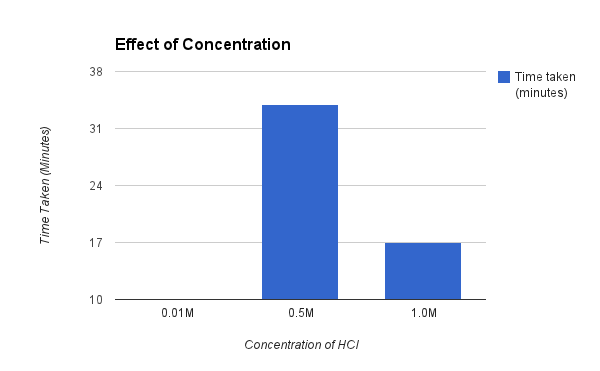
As seen on the graph below the highest the concentration of HCl (1.0) took only 17 minutes to dissolve the zinc metal where, on the opposite end of the scale, the lowest concentration of HCl (0.01) didn’t even have the ability to fully dissolve the piece of zinc metal. These results clearly agree with our hypothesis as when we raised the concentration it lowered the time taken to dissolve.
Overall the experiment, in my view, was a successful and valid experiment teaching me to understand the effect of concentration on the rate of reaction. If I were to replicate this experiment I would change the concentration of HCl as well as the material being dissolved as our timed result to the lowest concentration of HCl was not available. That would be on of our identified problems.
Conclusion
In my hypothesis I stated that If the rate at which a piece of zinc metal dissolves is related to the different concentrations of HCl, then the higher the concentration of HCl, the faster the rate at which the piece of zinc metal dissolves. In conclusion the outcome of this experiment directly related and agreed with my hypothesis as stated earlier in my discussion, the higher the concentration of the HCl resulted in lower amount of time taken to dissolve the zinc metal.
To test the effect of three different concentrations of HCl (0.1M, 1.0M, 2.0M) on the rate at which a piece of zinc metal dissolves.
Hypothesis
If the rate at which a piece of zinc metal dissolves is related to the different concentrations of HCl, then the higher the concentration of HCl, the faster the rate at which the piece of zinc metal dissolves.
Independent Variable: Concentration of HCl
Dependant Variable: Rate at which a piece of zinc metal dissolves
Controlled Variables: Amount of zinc metal
Beaker used
Amount of HCl
Materials
- 3 Test tubes
- Graduated Cylinder
- 5 mL of 0.01M Hydrochloric Acid
- 5 mL of .5M Hydrochloric Acid
- 5 mL of 1.0M Hydrochloric Acid
- 6 strips of zinc metal
- Funnel
- Safety Goggles
- Timer
Procedure:
1. Measure out 5 mL of 0.01M Hydrochloric Acid with the Graduated Cylinder and pour it into a test tube using a funnel
2. Drop 1 strip of zinc in the test tube and record the time it takes for the zinc metal to completely dissolve
3. Dispose the Hydrochloric Acid appropriately and rinse the test tube thoroughly
4. repeat steps 1-3 for the second trial
5. calculate the mean between the two trials.
6. Measure out 5 mL of 1.0M Hydrochloric Acid with the Graduated Cylinder and pour it into a test tube using a funnel.
7. Drop 1 strip of zinc in the test tube and record the time it takes for the zinc metal to completely dissolve.
8. Dispose the Hydrochloric Acid appropriately and rinse the test tube thoroughly.
9. Repeat steps 6-8 for the second trial
10. Calculate the mean between the two trials.
11. Measure out 5 mL of 2.0M Hydrochloric Acid with the Graduated Cylinder and pour it into a test tube using a funnel.
12. Drop 1 strip of zinc in the test tube and record the time it takes for the zinc metal to completely dissolve.
13. Dispose the Hydrochloric Acid appropriately and rinse the test tube thoroughly.
14.Repeat steps 11-13 for the second trial
15. Calculate the mean between the two trials.
Discussion
In my hypothesis I stated that If the rate at which a piece of zinc metal dissolves is related to the different concentrations of HCl, then the higher the concentration of HCl, the faster the rate at which the piece of zinc metal dissolves. My predictions were proved to be correct compared to the outcome of our experiments. These predictions could be hypothesized correctly due to the research done in the BC Science 10 textbook where it clearly states that if there is a greater concentration of reactant atoms and molecules present, there is a greater chance that collisions among them will occur.
In this experiment we tested the effect of concentration of HCl or Hydrochloric acid at which a piece of Zinc Metals dissolved. All control variables, a variable that remains unchanged or held constant to prevent its effects on the outcome of the experiment, were put forth and used as to find a result with no possible sources of error with a pure and unaltered result.
As seen on the graph below the highest the concentration of HCl (1.0) took only 17 minutes to dissolve the zinc metal where, on the opposite end of the scale, the lowest concentration of HCl (0.01) didn’t even have the ability to fully dissolve the piece of zinc metal. These results clearly agree with our hypothesis as when we raised the concentration it lowered the time taken to dissolve.
Overall the experiment, in my view, was a successful and valid experiment teaching me to understand the effect of concentration on the rate of reaction. If I were to replicate this experiment I would change the concentration of HCl as well as the material being dissolved as our timed result to the lowest concentration of HCl was not available. That would be on of our identified problems.
Conclusion
In my hypothesis I stated that If the rate at which a piece of zinc metal dissolves is related to the different concentrations of HCl, then the higher the concentration of HCl, the faster the rate at which the piece of zinc metal dissolves. In conclusion the outcome of this experiment directly related and agreed with my hypothesis as stated earlier in my discussion, the higher the concentration of the HCl resulted in lower amount of time taken to dissolve the zinc metal.