Purpose
To test the effect of three different temperatures of water at which an Alka Seltzer tablet dissolves
Hypothesis
If the rate at which an Alka Seltzer tablet dissolves is related to the different temperatures of water, then the higher the temperature of water, the faster the Alka Seltzer tablet will dissolve.
Independent variable: The temperature of water
Dependent variable: Rate at which the Alka Seltzer tablet dissolves
Controlled variables: Amount of water
Number of Alka Seltzer tablets
Beaker used
Materials:
Procedure:
1. Put 1 Alka Seltzer tablet in 100 mL of cold water and use a stopwatch to record the amount of time it takes for the Alka Seltzer tablet to dissolve.
2. Rinse the beaker with running water
3. Repeat Step 1 and 2 for the 2nd trial.
4. Calculate the mean between the two trials
6. Leave the hot plate on for 1 minute to let it heat up
5. Heat 100 mL of water on a hot plate for 1 minute
6. Take the beaker of hot water off of the hot plate using Heat Proof gloves
7. Put 1 Alka Seltzer tablet into the beaker of hot water and use a stopwatch to record the amount of time it takes for the Alka seltzer tablet to dissolve.
8. Rinse the beaker with running water
9. Repeat steps 5-8 for the 2nd trial
10. Calculate the mean between the results of the two trials
11. Heat 100 mL of water on a hot plate for 30 seconds
12.. Take the beaker of hot water off of the hot plate using Heat Proof gloves
13. Put 1 Alka Seltzer tablet into the beaker of warm water and use a stopwatch to record the amount of time it takes for the Alka Seltzer tablet to dissolve
14. repeat steps 10-13 for the 2nd trial.
15. Calculate the mean between the results of the two trials.
Discussion
In my hypothesis I stated that If the rate at which an Alka Seltzer tablet dissolves is related to the different temperatures of water, then the higher the temperature of water, the faster the Alka Seltzer tablet will dissolve. Similar to many commonly held beliefs and theories, this hypothesis was proven correct in that the higher the temperature resulted in a faster reaction rate. According to BC Science 10 one can use heat in order to increase the rate of reaction and vise versa.
Our experiments consisted of the careful testing of the effect of temperature on reaction rate ensuring to keep track of all control variables as to disallow any possible sources of error to occur. It was important for our class to partake in this experiment so that we may understand how temperature affects rate of reaction in our everyday lives. All Variables, most importantly the control variables, were not overlooked by any means in order to ensure correct results for a successful experiment.
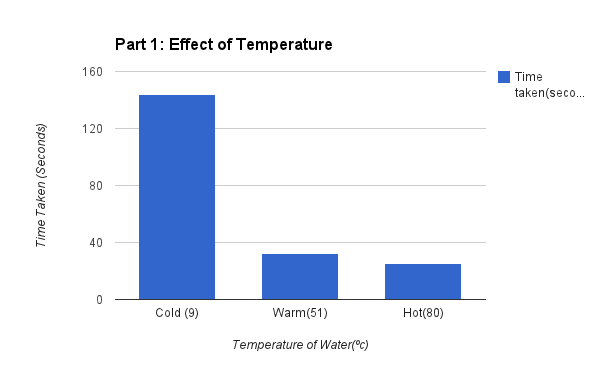
The results were as follows as well as shown above on the graph, cold water (9-degrees) dissolved the tablet in 144 seconds (on average), warm water (51-degrees) dissolved the tablet in 32 seconds (on average), and the warm water (81-degrees) dissolved the tablet in 25 seconds (on average). These results go to explain that the higher the temperature the faster the rate of reaction will occur.
The purpose of the experiment was to test the effect of three different temperatures of water at which an Alka Seltzer tablet dissolves. In my point of view this experiment was successful as it taught our group the effect of temperature in reaction rate. If this experiment were to take place again I would add more tests to record the reactions of different substances.
Conclusion
In my hypothesis i stated that If the rate at which an Alka Seltzer tablet dissolves is related to the different temperatures of water, then the higher the temperature of water, the faster the Alka Seltzer tablet will dissolve. The outcome of my experiment proved my hypothesis to be correct in that the hot water allowed for a much more rapid dissolve than that of the cold and warm water. I could predict this result because of the research done in the BC Science 10 textbook. Our lab consisted of the testing of how quickly an Alka Seltzer tablet dissolves in different temperatures of water. All variables were considered during our procedure as well as the controlled variables in order to ensure correct results for a successful experiment. As shown in the graph above where time is represented on the Y axis, the smallest bar (least time spent on average) was the warm water in that the Alka Seltzer tablet took only 25 seconds to dissolve compared to 144 seconds in the cold water. In my point of view this experiment was successful as it taught our group the effect of temperature in reaction rate. If this experiment were to take place again I would add more tests to record the reactions of different substances.
To test the effect of three different temperatures of water at which an Alka Seltzer tablet dissolves
Hypothesis
If the rate at which an Alka Seltzer tablet dissolves is related to the different temperatures of water, then the higher the temperature of water, the faster the Alka Seltzer tablet will dissolve.
Independent variable: The temperature of water
Dependent variable: Rate at which the Alka Seltzer tablet dissolves
Controlled variables: Amount of water
Number of Alka Seltzer tablets
Beaker used
Materials:
- 6 Alka seltzer tablets
- 100mL of 18ºc Cold Water
- 100mL 51ºc Warm water
- 100mL 80ºc Hot Water
- Glass Beaker
- Stopwatch
- Hot plate
- Heat Proof gloves
Procedure:
1. Put 1 Alka Seltzer tablet in 100 mL of cold water and use a stopwatch to record the amount of time it takes for the Alka Seltzer tablet to dissolve.
2. Rinse the beaker with running water
3. Repeat Step 1 and 2 for the 2nd trial.
4. Calculate the mean between the two trials
6. Leave the hot plate on for 1 minute to let it heat up
5. Heat 100 mL of water on a hot plate for 1 minute
6. Take the beaker of hot water off of the hot plate using Heat Proof gloves
7. Put 1 Alka Seltzer tablet into the beaker of hot water and use a stopwatch to record the amount of time it takes for the Alka seltzer tablet to dissolve.
8. Rinse the beaker with running water
9. Repeat steps 5-8 for the 2nd trial
10. Calculate the mean between the results of the two trials
11. Heat 100 mL of water on a hot plate for 30 seconds
12.. Take the beaker of hot water off of the hot plate using Heat Proof gloves
13. Put 1 Alka Seltzer tablet into the beaker of warm water and use a stopwatch to record the amount of time it takes for the Alka Seltzer tablet to dissolve
14. repeat steps 10-13 for the 2nd trial.
15. Calculate the mean between the results of the two trials.
Discussion
In my hypothesis I stated that If the rate at which an Alka Seltzer tablet dissolves is related to the different temperatures of water, then the higher the temperature of water, the faster the Alka Seltzer tablet will dissolve. Similar to many commonly held beliefs and theories, this hypothesis was proven correct in that the higher the temperature resulted in a faster reaction rate. According to BC Science 10 one can use heat in order to increase the rate of reaction and vise versa.
Our experiments consisted of the careful testing of the effect of temperature on reaction rate ensuring to keep track of all control variables as to disallow any possible sources of error to occur. It was important for our class to partake in this experiment so that we may understand how temperature affects rate of reaction in our everyday lives. All Variables, most importantly the control variables, were not overlooked by any means in order to ensure correct results for a successful experiment.
The results were as follows as well as shown above on the graph, cold water (9-degrees) dissolved the tablet in 144 seconds (on average), warm water (51-degrees) dissolved the tablet in 32 seconds (on average), and the warm water (81-degrees) dissolved the tablet in 25 seconds (on average). These results go to explain that the higher the temperature the faster the rate of reaction will occur.
The purpose of the experiment was to test the effect of three different temperatures of water at which an Alka Seltzer tablet dissolves. In my point of view this experiment was successful as it taught our group the effect of temperature in reaction rate. If this experiment were to take place again I would add more tests to record the reactions of different substances.
Conclusion
In my hypothesis i stated that If the rate at which an Alka Seltzer tablet dissolves is related to the different temperatures of water, then the higher the temperature of water, the faster the Alka Seltzer tablet will dissolve. The outcome of my experiment proved my hypothesis to be correct in that the hot water allowed for a much more rapid dissolve than that of the cold and warm water. I could predict this result because of the research done in the BC Science 10 textbook. Our lab consisted of the testing of how quickly an Alka Seltzer tablet dissolves in different temperatures of water. All variables were considered during our procedure as well as the controlled variables in order to ensure correct results for a successful experiment. As shown in the graph above where time is represented on the Y axis, the smallest bar (least time spent on average) was the warm water in that the Alka Seltzer tablet took only 25 seconds to dissolve compared to 144 seconds in the cold water. In my point of view this experiment was successful as it taught our group the effect of temperature in reaction rate. If this experiment were to take place again I would add more tests to record the reactions of different substances.