Purpose
To test the effect three different surface areas of an Alka Seltzer Tablet (whole tablet, chunks, crushed) on the rate at which an Alka Seltzer tablet dissolves.
Hypothesis
If the rate at which an Alka Seltzer tablet dissolves is related to the surface area than increasing the surface area will increase the rate at which the Alka Seltzer tablets dissolve faster.
Independent variable: rate at which the Alka Seltzer tablets dissolve
Dependent variable: surface area
Controlled variables: amount of water
amount of Alka Seltzer
temperature of water
Materials
Procedure
1. Put 1 whole Alka Seltzer Tablet into 100 mL of water and record the time it takes for it to dissolve
2. Rinse the beaker thoroughly.
3. Repeat steps 1 and 2 for 2nd trial
4. Calculate the mean between the two trials
5. Put 1 Alka Seltzer Tablet into a petri dish and break it into small pieces
6. Put the small pieces of Alka Seltzer into 100 mL of water and record the time it takes for it to dissolve. Be careful to put all bits and pieces into the beaker.
7. Rinse the beaker thoroughly
8. Repeat steps 5-7 for 2nd trial
9. Calculate the mean between the two trials.
10. Put 1 Alka Seltzer Tablet into a petri dish and completely crush it into powder.
11. Dump all the powder into 100 mL of water and record the time it takes for it to dissolve. Be careful to put all bits and pieces into the beaker.
12. Rinse the beaker thoroughly
13. Repeat steps 10-12 for 2nd trial.
14. Calculate the mean between the two trials.
Discussion
In my hypothesis I stated that If the rate at which an Alka Seltzer tablet dissolves is related to the surface area than increasing the surface area will increase the rate at which the Alka Seltzer tablets dissolve faster. My predictions were proved to be correct compared to the outcome of our experiments. These predictions could be hypothesized correctly due to the research done in the BC Science 10 textbook where it clearly states that the greater the surface area of the reactant (Alka Seltzer tablet) allows the particles of water to collide with the Alka Seltzer many more times per unit of time.
In this experiment we tested the effect of surface area of the rate at which Alka Seltzer dissolves in water. All control variables, a variable that remains unchanged or held constant to prevent its effects on the outcome of the experiment, were put forth and used as to find a result with no possible sources of error with a pure and unaltered result. For this experiment our control variables were the amount of water, the amount of Alka Seltzer and the temperature of the water
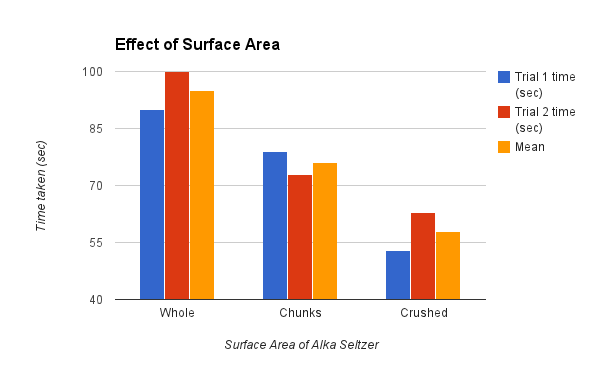
As seen on the graph below when the Alka Seltzer was crushed, in order to increase the surface area, the time at which it dissolved in water was visibly less than that of the crushed and whole Alka Seltzer tablets. With a mean, or average, difference of 38 seconds between the crushed and whole tablets the effect of surface area on the rate of reaction is quite obvious in the outcome of this experiment. This outcome could be easily predicted through the research of theoretical and expected results, as it is obvious that surface area does in fact have.
Overall the experiment, in my view, was a successful and valid experiment teaching me to understand the effect of surface area on the rate of reaction. If I were to replicate this experiment I would, as to speed up the process of testing, combine the surface area test with the temperature test as it would be interesting to see if the results are somewhat duplicated. I do not believe that our experiment was in any way affected or altered by any sources of error as we were aware and consciously kept all our control variables in check.
Conclusion
In my hypothesis I stated that if the rate at which an Alka Seltzer tablet dissolves is related to the surface area than increasing the surface area will increase the rate at which the Alka Seltzer tablets dissolve. In conclusion the outcome of this experiment directly relates to the research and previously believed scientific theories and explanations regarding this topic. My hypothesis was proved correct in that an increase of surface area resulted in a much faster rate of dissolve for the Alka Seltzer tablets.
To test the effect three different surface areas of an Alka Seltzer Tablet (whole tablet, chunks, crushed) on the rate at which an Alka Seltzer tablet dissolves.
Hypothesis
If the rate at which an Alka Seltzer tablet dissolves is related to the surface area than increasing the surface area will increase the rate at which the Alka Seltzer tablets dissolve faster.
Independent variable: rate at which the Alka Seltzer tablets dissolve
Dependent variable: surface area
Controlled variables: amount of water
amount of Alka Seltzer
temperature of water
Materials
- 6 Alka Seltzer Tablets
- Petri dish
- Mortar and pestel
- water
- beaker
- stopwatch
Procedure
1. Put 1 whole Alka Seltzer Tablet into 100 mL of water and record the time it takes for it to dissolve
2. Rinse the beaker thoroughly.
3. Repeat steps 1 and 2 for 2nd trial
4. Calculate the mean between the two trials
5. Put 1 Alka Seltzer Tablet into a petri dish and break it into small pieces
6. Put the small pieces of Alka Seltzer into 100 mL of water and record the time it takes for it to dissolve. Be careful to put all bits and pieces into the beaker.
7. Rinse the beaker thoroughly
8. Repeat steps 5-7 for 2nd trial
9. Calculate the mean between the two trials.
10. Put 1 Alka Seltzer Tablet into a petri dish and completely crush it into powder.
11. Dump all the powder into 100 mL of water and record the time it takes for it to dissolve. Be careful to put all bits and pieces into the beaker.
12. Rinse the beaker thoroughly
13. Repeat steps 10-12 for 2nd trial.
14. Calculate the mean between the two trials.
Discussion
In my hypothesis I stated that If the rate at which an Alka Seltzer tablet dissolves is related to the surface area than increasing the surface area will increase the rate at which the Alka Seltzer tablets dissolve faster. My predictions were proved to be correct compared to the outcome of our experiments. These predictions could be hypothesized correctly due to the research done in the BC Science 10 textbook where it clearly states that the greater the surface area of the reactant (Alka Seltzer tablet) allows the particles of water to collide with the Alka Seltzer many more times per unit of time.
In this experiment we tested the effect of surface area of the rate at which Alka Seltzer dissolves in water. All control variables, a variable that remains unchanged or held constant to prevent its effects on the outcome of the experiment, were put forth and used as to find a result with no possible sources of error with a pure and unaltered result. For this experiment our control variables were the amount of water, the amount of Alka Seltzer and the temperature of the water
As seen on the graph below when the Alka Seltzer was crushed, in order to increase the surface area, the time at which it dissolved in water was visibly less than that of the crushed and whole Alka Seltzer tablets. With a mean, or average, difference of 38 seconds between the crushed and whole tablets the effect of surface area on the rate of reaction is quite obvious in the outcome of this experiment. This outcome could be easily predicted through the research of theoretical and expected results, as it is obvious that surface area does in fact have.
Overall the experiment, in my view, was a successful and valid experiment teaching me to understand the effect of surface area on the rate of reaction. If I were to replicate this experiment I would, as to speed up the process of testing, combine the surface area test with the temperature test as it would be interesting to see if the results are somewhat duplicated. I do not believe that our experiment was in any way affected or altered by any sources of error as we were aware and consciously kept all our control variables in check.
Conclusion
In my hypothesis I stated that if the rate at which an Alka Seltzer tablet dissolves is related to the surface area than increasing the surface area will increase the rate at which the Alka Seltzer tablets dissolve. In conclusion the outcome of this experiment directly relates to the research and previously believed scientific theories and explanations regarding this topic. My hypothesis was proved correct in that an increase of surface area resulted in a much faster rate of dissolve for the Alka Seltzer tablets.